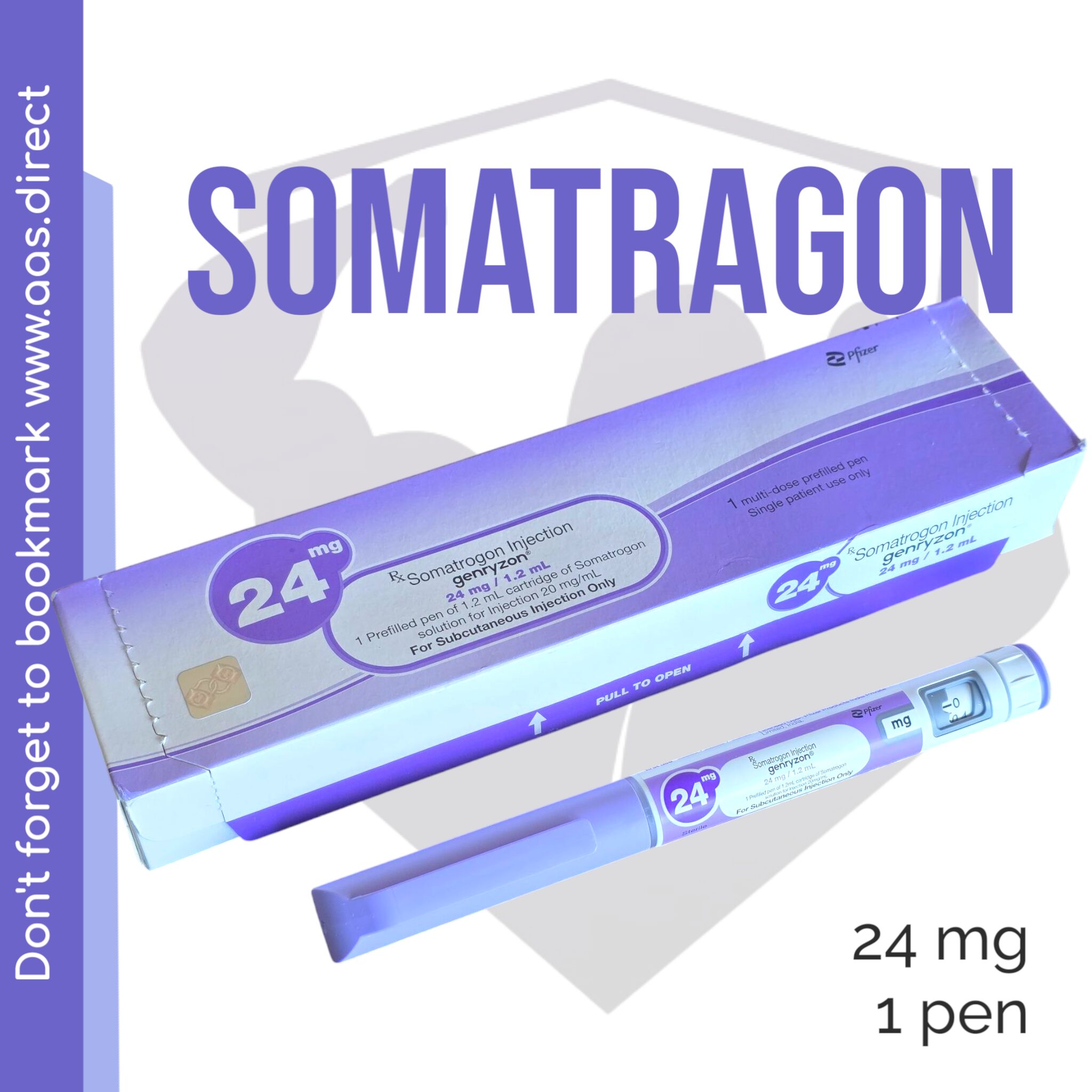
Genryzon (Somatrogon) 24mg – Weekly Growth Hormone Optimization
Genryzon (Somatrogon) is a next-generation, long-acting recombinant human growth hormone (rhGH) analog fused with C-terminal peptide extensions to increase its half-life. Designed for once-weekly subcutaneous injection, Genryzon delivers steady and elevated GH and IGF-1 levels without the peaks and valleys of daily growth hormone therapy.
Each vial contains 24mg of lyophilized Genryzon (Somatrogon), ideal for advanced users seeking a long-acting, clinically proven growth hormone alternative for fat loss, muscle gain, recovery, and anti-aging benefits.
 Key Benefits
Key Benefits
- Stimulates IGF-1 Production: Promotes cellular growth, muscle repair, and regeneration
- Accelerates Fat Loss: Enhances lipolysis and metabolic rate
- Increases Lean Mass: Improves muscle density and nitrogen retention
- Improves Sleep & Recovery: Optimizes healing during rest periods
- Longevity Support: May improve skin, joints, cognition, and quality of life
 How Genryzon (Somatrogon) Works
How Genryzon (Somatrogon) Works
Somatrogon is a bioengineered fusion protein combining recombinant human growth hormone (rhGH) with segments of human chorionic gonadotropin and albumin-binding domains. These extensions delay renal clearance and extend the peptide’s half-life to 6+ days — allowing for convenient once-weekly dosing while maintaining consistent GH activity. This improves IGF-1 regulation and reduces injection frequency compared to traditional GH protocols.
Product Specifications
| Specification | Details |
|---|---|
| Peptide | Somatrogon (Genryzon) |
| Strength | 24mg prefilled pen |
| Dosing Schedule | Once per week, 0.2–0.6mg/kg (adjust based on goals) |
| Cycle Duration | 12–24 weeks (long-term support) |
| Half-Life | ~6 days |
| Storage | Refrigerate after mixing |
| Reconstitution | premixed |
Advanced Use & Stacking – Growth Hormone (GH) Optimization
Synergistic Stacks for Maximum Benefits
- With Ipamorelin or CJC-1295: Enhances pituitary function and promotes stronger GH pulsatility, supporting lean mass, recovery, and anti-aging effects.
- With NAD+ or MOTS-c: Supports cellular repair, mitochondrial function, and energy production, complementing GH-mediated tissue regeneration.
- With Metformin or Tirzepatide: Improves insulin sensitivity while boosting fat loss, optimizing metabolic balance during GH-focused protocols.
Why Choose Genryzon?
- Convenient once-weekly injection schedule for consistent plasma GH levels
- Stable GH levels without suppression or rebound effects
- Clinically studied recombinant analog with a long safety record
- Ideal for growth, fat loss, recovery, and longevity optimization
- Can be safely stacked with compounds like Testosterone Cypionate, Testosterone Enanthate, Nandrolone Decanoate, or MK-677/Ibutamoren for enhanced anabolic and recovery benefits
Warnings & Considerations
- Do not use during pregnancy or with active malignancy
- Monitor IGF-1 levels during extended use
- Consult a healthcare provider before starting any GH-based research protocol
- Intended for research and laboratory purposes; not for human or veterinary use
Ideal For
- Fitness enthusiasts seeking lean mass and improved recovery
- Men and women exploring anti-aging or longevity therapies
- Individuals with GH deficiency or age-related decline
- Those wanting the benefits of GH without daily injections
This version naturally integrates your AASDirect links and targets keywords like:
- “Growth hormone research chemical”
- “Genryzon GH optimization”
- “GH stack for fat loss and lean mass”
- “Ipamorelin CJC-1295 stack”
- “MK-677 GH synergy”



Reviews
There are no reviews yet.